重磅分享!清洁验证风险评估指南!
校对:流浪的沙子
GMP办公室翻译组组织翻译,欢迎加入GMP办公室翻译组QQ群参与更多指南翻译!
译自2020年8月份发布的《ISPE基准指南:清洁验证生命周期–应用,方法和控制(ISPE Baseline Guide: Cleaning Validation Lifecycle: Applications, Methods, and Controls)》
3 Risk Management
清洁验证风险管理
3.1 RiskManagement Description Overview and Regulatory Expectations
3.1风险管理概览和监管要求
Quality RiskManagement (QRM) is a rational approach to enable good decisions. Cleaningvalidation, like other GMP validation activities, is not exempted from theregulatory expectation of using risk management to control potential hazards,reduce risks, and establish sound cleaning processes. In fact, a risk-basedcleaning validation strategy with justifiable and achievable acceptancecriteria is crucial in attaining a compliant cleaning validation program.
质量风险管理(QRM)是实现良好决策的合理方法。正如其他GMP验证活动一样,清洁验证也需要使用风险管理来控制潜在风险,降低风险,并建立合理的清洁工艺。实际上,基于风险的清洁验证策略以及具有合理且可实现的接受标准,对于获得合规的清洁验证程序至关重要。
Forexample, validation master plans for cleaning should either start with adocumented risk assessment exercise or at least include the risk assessmentprocess in the early stages of developing a new program. In today’s pharmaceuticalregulatory landscape, a retrospective risk assessment is not only a goodpractice, it is expected within cleaning programs, even if that program hasbeen grandfathered as acceptable.
例如,清洁验证主计划应从书面的风险评估活动开始,或者至少在制定新计划的早期阶段就包括风险评估过程。在当今的药品监管环境中,回顾性(倒推式)风险评估不是一种好的做法,而应在清洁计划中采用,尽管历史上它被认为是可以接受的。
3.1.1Regulatory Expectations
监管要求
Quality riskmanagement is a regulatory expectation, as noted in FDA’s Pharmaceutical CGMPsfor the 21th Century [18], which aims to encourage implementation of risk-basedapproaches that focus on critical areas for maintaining or improving productquality.
正如FDA面向21世纪的药品CGMP[18]所指出的那样,质量风险管理是一项监管期望,其目的是鼓励实施基于风险的方法,关注于维持或改善产品质量的关键领域。
Expectationsfor the application of QRM principles to validation are clearly stated inregulatory documents.
法规文件中清楚地说明了QRM原则应用于验证的期望。
For example,EudraLex Annex 15 [4] states applications for QRM in validation:
例如,EudraLex附录15[4]说明了QRM在验证中的应用:
Todetermine scope and extent of qualification and validation.
确定确认和验证的范围和程度。
To reassessrisks after gaining more knowledge from commercial production.
从商业生产中获得更多的知识重新评估风险。
Todetermine criticality of process parameters.
确定关键工艺参数。
To evaluateplanned changes to determine potential impact.
评估计划性变更以确定潜在的影响。
To justify bracketing approaches.
论证括号法的合理性。
To determine the variable factors which influence cleaningeffectiveness and performance.
确定影响清洁效果和性能的可变因素。
To justifyselected cleaning limits.
证明所选择清洁限度的合理性。
To justifythe number of times the cleaning procedure should be executed (number of runs)for validation.
证明清洁程序验证时执行次数的合理性。
Todetermine the risks presented by microbial and endotoxin contamination duringthe development of cleaning validation protocols.
清洁验证方案开发过程中,确定微生物和内毒素污染带来的风险。
Similarly,the FDA Guidance for Industry: Process Validation [5] calls for QRM principlesto be applied:
同样,FDA《行业指南:工艺验证[5]》要求遵循的QRM原则:
Todetermine the degree of controls needed to control process variation.
确定控制工艺变化所需的程度。
To screenpotential variables for Design of Experiment (DOE) studies to minimize thetotal number of experiments conducted while maximizing knowledge gained.
筛选实验设计(DOE)研究的潜在变量,以使实验次数最少,而能最大限度地获得知识。
To supportthe prioritization of certain equipment qualification activities and toidentify the level of effort needed in both the performance and documentationof qualification activities.
支持对某些设备确认活动的优先级,并确定确认活动的性能和文档水平。
To justifythe sampling location, sampling frequency, and confidence level for samplingplans.
证明取样点,取样频次和取样计划置信水平的合理性。
Todetermine criteria for process performance indicators.
确定工艺性能指标的标准。
Furthermore,EMA in its guidance for industry to set HBELs recognizes that cleaning is arisk reducing measure.
“A more scientific case by case approach iswarranted for risk identification and to support risk reduction measures forall classes of pharmaceutical substances.” [11]
此外,EMA在其HBEL行业指南中亦指出清洁是降低风险的措施。“对于所有级别的药用物质,有必要采取更科学的个案分析方法来识别风险,并支持风险降低措施。” [11]
Inaddition, once a health-based assessment has been completed, the _width="-30px" src="https://st.yunmeow.com/20251202/5143bcf2-2185-47c2-8c2b-6db186ac96f5.png">
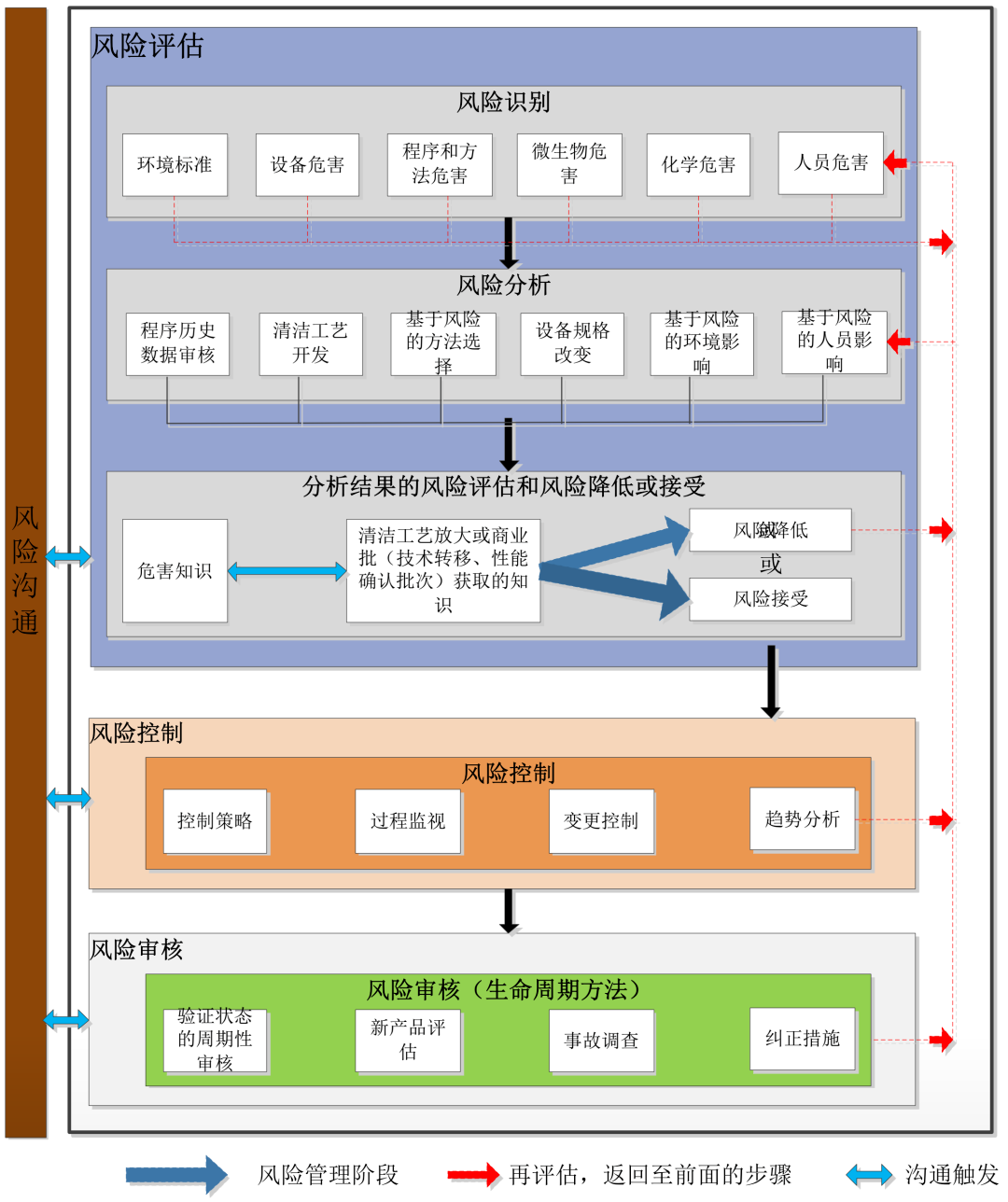
There are many tools available to support theapplication of QRM principles. The most common are:
有许多工具可以用来支持质量风险管理原则的应用。最常见的有:
Failure Mode Effects Analysis (FMEA)
失效模式影响分析(FMEA)
Fault Tree Analysis (FTA)
故障树分析
Hazzard Analysis and Critical Control Points(HACCP)
危害分析和关键控制点(HACCP)
Fishbone/Ishikawa
鱼骨图/石川图
One hint to secure support between Subject Matter Experts (SMEs) andcolleagues during risk assessments is to simplify (when possible) the logicalpath from supporting _width="-30px" src="https://st.yunmeow.com/20251202/d3548b0a-6784-4487-a2d6-360110d9ae76.png">
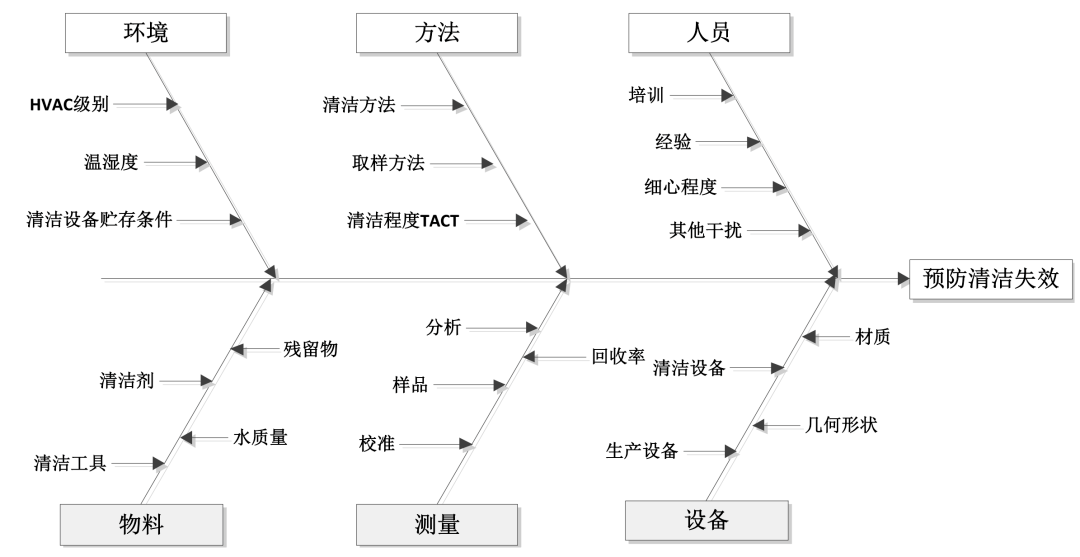
Adapted fromISPE Training Slides [40]
改自ISPE 培训PPT[40]
3.2 Risk ManagementApplied to the Cleaning Validation Program
风险管理在清洁验证程序中的应用
Riskmanagement for cleaning processes focuses on the hazard represented by theactive and chemical residues on product contact surfaces, the impact orseverity represented by such residues, the likelihood of these residues being present,and the ability to detect them.
清洁工艺的风险管理重点在于产品接触表面上的活性残留物和化学残留物所带来的危害,此类残留物所带来的影响或严重性,存在这些残留物的可能性以及对其进行检测的能力。
Manycleaning regimens originally focused on visual cleanliness as a measure of success.However, there are multiple sources of hazards in a cleaning process that needto be well understood in order to guide the planning and decision making fordeveloping effective and compliant cleaning processes.
许多清洁方案最初都将目视清洁作为成功的标准。但是,清洁过程中存在多种危害源,需要充分了解这些危害,以指导制定计划和决策来开发有效且合规的清洁工艺。
One ofthe most important hazards in a cleaning program comes from the type of soilsto be cleaned. Some chemicals and actives present low risks to patients andothers higher risks, as represented in Figure 3.3.
在清洁程序中最重要的危害之一是待清洁的污染类型。如图3.3所示,某些化学药品和活性物质对患者的风险较低,而有些则较高。
Figure3.3: Hazard Continuum [3]
图3.3:危害轴[3]
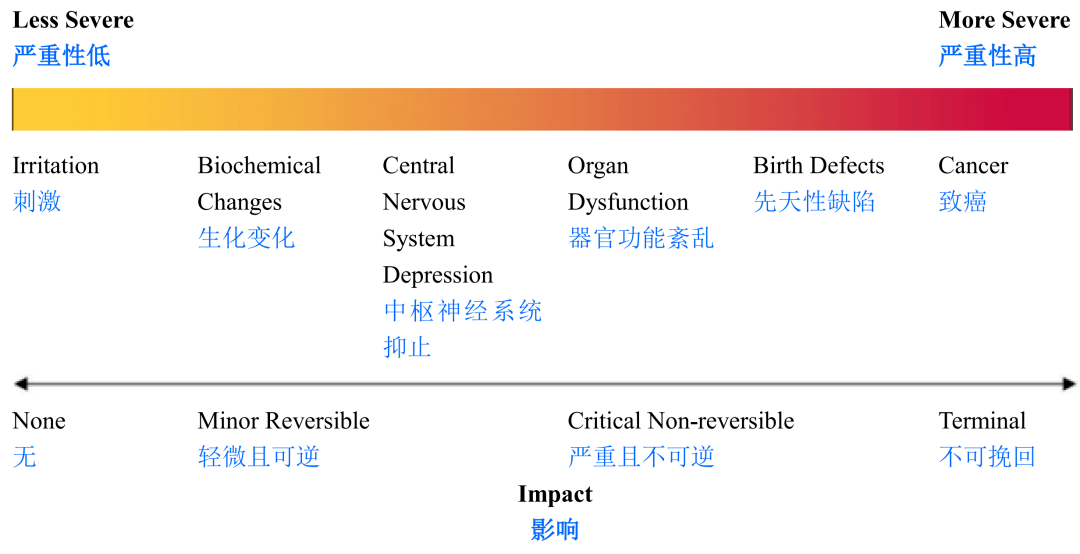
Therecommended approach for protecting patients from chemical and active residueis to use toxicological cellpadding="0">
Cleaning Validation Milestone
清洁验证里程碑
Tasks
任务
Considerations
考虑点
Validation Master Plan
验证主计划
Matrix approach or testing all products (multiproduct facility)
矩阵方式或测试所有的产品(多产品共用设施)
Define “worst-case” approach for validating products and API
定义清洁验证“最差条件”产品和API
Define sampling plan
定义取样计划
Sampling methods, locations, frequency, and confidence levels
取样方法,取样点,频次和置信水平
Selection of analytical methods
分析方法选择
HPLC versus Total Organic Carbon (TOC) along with appropriate microbial testing techniques, LOD, method variability
HPLC与总有机碳(TOC)以及适当的微生物检测技术,检测限(LOD),方法变异性
Design and Development
设计和开发
Determine target HBEL and perform risk assessment to evaluate cleaning
limits
确定目标HBEL、进行风险评估以评估清洁限度
Availability of PDE/ADE or clinical>
Determine parameters for the cleaning process
确定清洁工艺参数
Justify bracketing and grouping approaches
论证括号法和分组法
Determine interactions between parameters and justify best ranges for effectiveness and performance
确定参数之间的相互作用,并证明有效性和性能的最佳范围
Qualification (PPQ)
确认(PPQ)
Test equipment hard to clean areas to confirm CV
测试设备难以清洁区域以证实清洁验证
Confirm equipment is sufficiently clean to complete CV through appropriate testing and sampling locations
通过适当的测试和取样位置确认设备得到充分清洁以完成清洁验证
Determine number of runs for cleaning qualification
确定清洁确认执行的次数
Optimum number of times cleaning procedures must be executed to cover the scope of cleaning validation
清洁程序应被执行,且能覆盖清洁验证范围
Verification (CPV)
确证(CPV)
Find areas where the processes can be improved
寻找可以改进的过程
CHT studies or reduced testing strategies during campaigning production, evaluation of current hazards to update controls
在生产活动期间进行洁净保持时间(CHT)研究或减少测试策略,评估当前危害以更新控制措施
Ongoing Monitoring
持续监测
Ensure that cleaning limits remain appropriate and that periodic testing satisfies requirements
确保清洁限度保持适当,并且定期测试结果满足要求
Additional process knowledge could change initial PDE/ADE calculations; verification via spot checking the current hazards can reduce risk
额外的工艺知识可能会改变初始的PDE /ADE计算;通过对当前危害的抽查确认风险可以降低
Evaluate opportunities for reduced testing
评估减少测试的机会
Level of controls (testing) should be commensurate to current level of risks
控制(测试)的水平应与当前的风险水平相适应。
Periodic Review
定期审查
Assemble a cross-functional team for assessment to evaluate and report on the cleaning validation program effectiveness
组件一个跨职能团队以评估和报告清洁验证计划的有效性
This report should be available for regulatory review to highlight updates in cleaning validation control strategies
此报告应在法规检查期间可用,应突出清洁验证控制策略的更新
Change Management
变更管理
Evaluate planned changes to determine potential impact
评估计划性变更以决定潜在的影响
Consider impact to validation control plan.
考虑对验证控制计划的影响
Consider accumulative impact of changes.
考虑变更累计的影响
Additional risk evaluation applications are discussed inSections 3.2.1 to 3.2.4 using the cleaning validation lifecycle.
使用清洁验证生命周期在3.2.1至3.2.4节中讨论了其他风险评估应用。
3.2.1Initial Cleaning Validation Assessments
3.2.1初始清洁验证评估
Validation assessments at this stage help determine the scopeand extent of qualification and validation efforts from a perspective of risk.
此阶段的验证评估有助于从风险的角度确定验证工作的范围和程度。
The assessment conclusion should recommend validation,verification (i.e., dedicated indirect equipment), or no validation (i.e.,waste collection vessels) requirements. The assessment should also identify theextent of the effort (number of runs or products involved, studies anticipatedto be needed, special training considerations, etc.). Table 3.2 provides a listof areas to consider during assessments for validation.
评估结论应建议验证,确认(例如,专用的非直接接触设备)或不需验证(如废弃物收集容器)的要求。评估还应确定工作的程度(涉及的运行次数或产品数量,预期需要的研究,特殊的培训等)。表3.2列出了评估中需要考虑的领域。
Table 3.2: Validation Assessment Elements for Cleaning
表3.2:清洁验证评估要素
Scope 范围 | Evaluation Scope 评估范围 |
Analytical Methods 分析方法 | · Supplies (e.g., containers, swabs) 供应商(如容器,拭子) · Specificity 专属性 · Robustness 耐用性 · Range 范围 · Sensitivity 灵敏度 · Active degradation 活性物质降解 · Procedure for unknown peaks 处理未知峰的程序 |
Cleaning Agents 清洁剂 | · ADE/PDE levels ADE/PDE水平 · Cleaning effectiveness 清洁的效果 · Rinsability 漂洗力 · Detectability 可检测性 |
Cleaning Cycle Development 清洁周期开发 | · Number of runs 运行次数 |
Cleaning Procedure 清洁程序 | · TACT* characteristics TACT(时间,作用,化学和温度)特性 · Reproducibility (e.g., automated, semiautomated, manual) 重现性(自动,半自动,手动) · Level of training 培训水平 · Level of detail (e.g., manual cleaning) 详细程度(如手动清洁) |
Cleaning Qualification Execution 清洁确认执行 | · Number of runs 次数 |
Hold Times 保持时间 | · Dirty (e.g., effect on cleanability) 脏的保持时间(如对可清洁性的影响) · Clean (e.g., length, storage, proliferation) 洁净保持时间(如时长、存放条件,增殖) |
Manufacturing Equipment 生产设备 | · Equipment and process product contact points 设备和工艺产品的接触点 · Equipment complexity (e.g., components, circuits, shape) 设备的复杂性(如组件、回路、形状等) · Buildup concerns 累积相关 · Grouping (e.g., products, equipment) 分组(如产品、设备) |
Manufacturing Process 制造过程 | · Soil cleanability 污染可清洁性 · Process step criticality 工艺步骤的关键性 · Soil load/batch size variation 污染负荷/批量变化 |
Residue 残留 | · ADE/PDE levels of process residues (e.g., APIs, excipients, degradants, process aides, lubricants) 过程残留物(例如,API、辅料、降解产物、工艺助剂、润滑剂)的ADE/PDE水平 · Cleanability 可清洁性 · Detectability 可检测性 |
Safety Cleaning Limits 清洁限度安全性 | · Cleaning limit calculation and justification 清洁限度计算和论证 · Rationale for chosen cleaning limit method 选择清洁限度方法的原理 · HBEL safety limit calculation and justification HBEL安全限度计算和论证 |
Sampling Approach 取样方法 | · Indirect (e.g., timing, sequence (in-line, grab sample, separate)) 间接取样(时间,次序(在线,擦拭,单独)) · Direct (e.g., accessibility, locations) 直接取样(如可及性,取样位置) · Visual inspection (e.g., accessibility, lighting) 目视检查(如可及性,照明) |
Sampling Recovery 取样回收 | · Recoverability (cleaning agent, active ingredient, and microbial) 可回收性(清洁剂,活性物质和微生物) · Materials of construction 结构材质 · Percent recovery 回收百分比 |
*TACT = Time, Action, Chemical, and Temperature TACT=时间,作用,化学,温度 |
3.2.2 Introduction of New ProductsRisk Assessments
3.2.2新产品引入的风险评估
Introducing new products into afacility requires in-depth knowledge of the new hazards this may imply. When assessingthe validation effort for legacy cleaning processes, the assessment shouldconsider factors such as:
将新产品引入工厂需要深入了解新的未知危害。在评估已有清洁过程的验证工作时,应考虑以下因素:
Historical knowledge of current processes
当前工艺的历史知识
Process and equipment similarities
工艺和设备的相似性
Cleaning methods used
使用的清洁方法
Cleaning agents used
使用的清洁剂
Equivalent or worst-caseexcipients/formulations
等效或最差情况的辅料/制剂
If the new product to be introduced does not represent a newworst-case chemical or active to clean, then the existing cleaning process maybe considered sufficient. A comprehensive risk assessment is completed toevaluate any changes to the existing control plans, including updatingjustification reports for HBEL, sampling, and all other elements of thecleaning program. Conversely, if the new product introduces new chemicals oractives that exceed the capability of the current cleaning process, then acomprehensive evaluation of the cleaning program is required.
如果拟引入的新产品不会引起新的最差情况化学物质或活性物质,则可以认为现有的清洁工艺已足够。应完成一份全面的风险评估报告,以评估对现有控制计划的任何更改,包括更新有关HBEL,取样和清洁计划所有其他要素的论证报告。相反,如果新产品引入的新化学品或活性物质超出了当前清洁工艺的能力,则需要对清洁程序进行全面评估。
Refer to Appendix 8 for a case studyapplying QRM principles to the introduction of new products in an existingfacility.
有关将QRM原理应用于现有工厂中引入新产品的案例研究,请参阅附录8。
3.2.3 Ongoing Monitoring Maintenance Risk Assessments
3.2.3持续监测维护风险评估
The level, type, and frequency of testing during the ongoingmonitoring of a validated cleaning process is evaluated via an initial riskassessment. This ensures the implementation of a monitoring program that isscience and riskbased. Additionally, as risk is reduced, the level ofmonitoring can be reduced.
通过初始风险评估,评估对已验证清洁工艺进行持续监测的测试级别,类型和频率。这样可以确保实施基于科学和风险的监测程序。另外,随着风险的降低,监测级别也可以降低。
The scope of the risk assessment should be determined early inthe process, ideally as part of the Validation Master Plan (VMP) and periodicreview of cleaning processes.
风险评估的范围应在过程的早期确定,理想情况下应作为验证主计划(VMP)和清洁工艺定期审查的一部分。
3.2.4 Routine Operation of Cleaning Process Risk Assessments
3.2.4清洁工艺风险评估的日常操作
As part of the ongoing verificationof cleaning processes, a risk assessment is performed to assess the cleaning processfor changes in hazards and corresponding risks. It assesses changes inprocesses, procedures, activities, and determines points of highest impact. Theassessment should be a living document to be updated on a periodic schedule(i.e., during cleaning periodic reviews) or be part of a control strategy tocapture changes to the process.
作为清洁工艺持续确认的一部分,应执行风险评估,以评估清洁工艺中的危害变化和相应的风险。它评估工艺,程序,活动的变更,并确定影响最大的点。该评估应是一份定期更新的动态文件(例如,在清洁定期审查期间),或作为控制策略的一部分,以捕获过程的变化。
HACCP or modified versions are goodfor assessing the process and determining potential failure points or weak pointswithin the process.
HACCP或其改良版本非常适合评估工艺并确定工艺中潜在的失败点或薄弱点。
Considerations:
考虑点:
Changes documented as part of thecleaning process periodic review
作为清洁工艺定期审查的一部分记录的变更
In-process sampling methods
过程中采样方法
Process performance
工艺性能
Changes in manufacturing processsteps
生产工艺步骤的变更
Changeover procedures
切换程序
Training program
培训计划
Changes in CIP/COP recipes andalarms
CIP /COP配方的更改和报警
New product and equipment grouping
新产品和设备分组
Equipment maintenance
设备维护保养
3.2.5 Additional Applications
3.2.5其他应用
For complex processes or scenarios, separate risk assessmentscan be used to evaluate special hazards and identify appropriate controlmeasures to mitigate risks. For example:
对于复杂的过程或场景,可以使用单独的风险评估来评估特定危害并确定适当的控制措施以减轻风险。例如:
Implementing an aggressive cleaning validation groupingstrategy
实施积极的清洁验证分组策略
Using reduced Material of Construction (MOC) recoverygroupings to represent larger groups of materials
使用代表性的结构材质(MOC)回收组来代表大量的材质组
Optimizing the level of detail for non-critical manualcleaning activities
优化非关键手动清洁活动的详细程度
These scenarios require that SMEs understand how to effectivelyanalyze and control potential hazards. The output of these risk assessments isused to support better decisions.
这些情况要求SME了解如何有效分析和控制潜在危害。风险评估的结果用于支持做出更好的决策。